Afrezza Inhaled Insulin Now Available in U.S. Pharmacies Nationwide
| Marco Foronda | | Feb 03, 2015 10:11 AM EST |
Afrezza
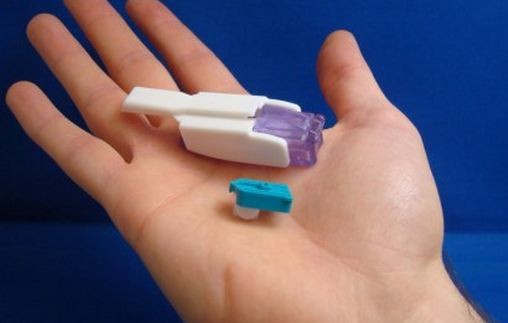
Sanofi and MannKind Corp. announced Tuesday that Afrezza (insulin human) Inhalation Powder, the only inhaled insulin, is now available by prescription at U.S. retail pharmacies nationwide.
The U.S. Food and Drug Administration approved Afrezza for use by adults with type 1 and type 2 diabetes. Afrezza uses a whistle-sized inhaler and was developed in competition against Pfizer's rival Exubera.
Like Us on Facebook
Afrezza is a drug-device combination product that consists of a dry formulation of human insulin delivered from a small and portable inhaler. It helps patients achieve blood sugar control. It can be rapidly absorbed and has a short duration of action. It is administered at the start of meal.
Afrezza manages to control high blood sugar as part of a diabetes management plan that may include diet, exercise and other diabetes medications.
Experts warned Afrezza should not be used in patients with chronic lung disease like asthma or COPD and can't be used to treat diabetic ketoacidosis. It's also not recommended for smokers or recent ex-smokers. Both companies advised people not to use Afrezza as an alternative for long-acting insulin.
Sanofi and MannKind entered into a worldwide exclusive licensing agreement to develop and commercialize Afrezza. As per the detailed agreement, Sanofi is responsible for global commercial, regulatory and development activities.
Afrezza costs US$7.54 for a daily dose of 12 units. It's pricier than US$3.14 Apidra, which is the injectable equivalent in Sanofi's drugs stable.
TagsSanofi, MannKind, Afrezza, insulin, Inhaled Insulin, Diabetes, type 1 diabetes, Type 2 diabetes, USFDA, medical drug
©2015 Chinatopix All rights reserved. Do not reproduce without permission
EDITOR'S PICKS
-

Did the Trump administration just announce plans for a trade war with ‘hostile’ China and Russia?
-

US Senate passes Taiwan travel bill slammed by China
-

As Yan Sihong’s family grieves, here are other Chinese students who went missing abroad. Some have never been found
-

Beijing blasts Western critics who ‘smear China’ with the term sharp power
-

China Envoy Seeks to Defuse Tensions With U.S. as a Trade War Brews
-

Singapore's Deputy PM Provides Bitcoin Vote of Confidence Amid China's Blanket Bans
-

China warns investors over risks in overseas virtual currency trading
-

Chinese government most trustworthy: survey
-

Kashima Antlers On Course For Back-To-Back Titles
MOST POPULAR
LATEST NEWS
Zhou Yongkang: China's Former Security Chief Sentenced to Life in Prison

China's former Chief of the Ministry of Public Security, Zhou Yongkang, has been given a life sentence after he was found guilty of abusing his office, bribery and deliberately ... Full Article
TRENDING STORY

China Pork Prices Expected to Stabilize As The Supplies Recover

Elephone P9000 Smartphone is now on Sale on Amazon India

There's a Big Chance Cliffhangers Won't Still Be Resolved When Grey's Anatomy Season 13 Returns

Supreme Court Ruled on Samsung vs Apple Dispute for Patent Infringement

Microsoft Surface Pro 5 Rumors and Release Date: What is the Latest?